[author: Donald Zuhn]
Earlier this year, the Intellectual Property Owners Association (IPO) released its 29th annual list of the top 300 organizations receiving U.S. patents. As in past years, Patent Docs used the IPO's list of top patent holders to compile a list of the top life sciences companies and organizations receiving U.S. patents in 2011 (see "IPO Releases List of Top 300 Patent Holders for 2011"). As noted in the IPO's Top 300 list, 244,430 patents were issued in 2011, which constituted a 4.6% increase over the 233,127 patents that issued in 2010. While the number of biotech/pharma patents also increased in 2011, the jump was not quite as steep, with 19,138 biotech/pharma patents being issued in 2011 as compared with the 18,900 biotech/pharma patents that issued in 2010 (1.3% increase).
This increase in biotech/pharma patents is shown in the chart below, which also shows the number of biotech/pharma patents that issued between 2002 and 2012 (blue bars, left axis) and the number of biotech/pharma applications that published (red line, right axis) over this same time frame (note: figures for 2012 are projected based on issuances and publications through today).
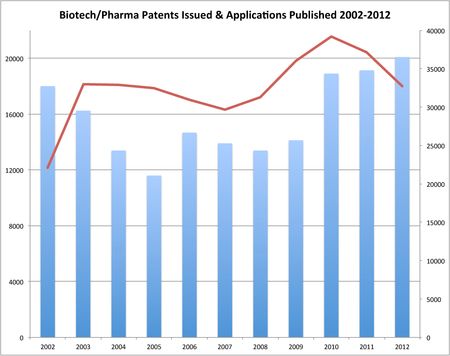
In the chart above, the blue bars represent the number of patents that issued in the specified year and which were designated as belonging to the following classes (each having an assigned art unit in Technology Center 1600, which encompasses biotechnology and organic chemistry): 260 (Chemistry of Carbon Compounds), 424 (Drug, Bio-Affecting and Body Treating Compositions), 435 (Chemistry: Molecular Biology and Microbiology), 436 (Chemistry: Analytical and Immunological Testing), 504 (Plant Protecting and Regulating Compositions), 506 (Combinatorial Chemistry Technology: Method, Library, Apparatus), 514 (Drug, Bio-Affecting and Body Treating Compositions), 518 (Chemistry: Fischer-Tropsch Processes; or Purification or Recovery of Products Thereof), 530 (Chemistry: Natural Resins or Derivatives; Peptides or Proteins; Lignins or Reaction Products Thereof), 532, 534, 536, 540, 546, 548, 549, 552, 554, 556, 558, 560, 562, 564, 568, 570 (Organic Compounds -- Part of the Class 532-570 Series), 730 (Data Processing: Structural Design, Modeling, Simulation, And Emulation), 800 (Multicellular Living Organisms and Unmodified Parts Thereof and Related Processes), and/or 930 (Peptide or Protein Sequence), 987 (Organic Compounds Containing a Bi, Sb, As, or P Atom or Containing a Metal Atom of the 6th to 8th Group of the Periodic System). The red line represents the number of patent applications designated as belonging to one of the above classes that published in the specified year (because the AIPA mandated publication of certain U.S. applications filed after November 28, 2000, only 22,097 biotech/pharma applications were published in 2002).
If the numbers of patents issued and applications published to date hold up during the remainder of the year, biotech/pharma patent issuances would be expected to rise to 20,087 in 2012. However, published biotech/pharma applications would be expected to drop from 37,125 in 2011 to 32,738. The expected increase in issued biotech/pharma patents and corresponding decrease in published biotech/pharma applications would continue a trend that began in 2010.