[author: Kevin E. Noonan]
 A three judge Federal Circuit panel went a long way towards disentangling its jurisprudence on the question of obviousness-type double patenting, in affirming a District Court finding that the doctrine did not apply to the patent claims at issue in Eli Lilly & Co. v. Teva. In what seems to becoming a penchant of this Court (see "Momenta Pharmaceuticals Inc. v. Amphastar Pharmaceuticals, Inc.: 'The Rest of the Story'"), the panel distinguished its decision rather than simply finding the Court's most recent spate of decisions to have gone astray (see "Cert. Denied in Eli Lilly v. Sun Pharmaceuticals").
A three judge Federal Circuit panel went a long way towards disentangling its jurisprudence on the question of obviousness-type double patenting, in affirming a District Court finding that the doctrine did not apply to the patent claims at issue in Eli Lilly & Co. v. Teva. In what seems to becoming a penchant of this Court (see "Momenta Pharmaceuticals Inc. v. Amphastar Pharmaceuticals, Inc.: 'The Rest of the Story'"), the panel distinguished its decision rather than simply finding the Court's most recent spate of decisions to have gone astray (see "Cert. Denied in Eli Lilly v. Sun Pharmaceuticals").
The present case concerns pemetrexed, a folate metabolism inhibitor synthesized by Professor Ed Taylor at Princeton University and developed by Eli Lilly & Co., sold as the anticancer drug Alimta® for treating mesothelioma and small cell lung cancer. Pemetrexed's biological activity, as with earlier folate inhibitors like methotrexate, is understood to result from its structural similarity to naturally occurring folic acid, which is needed for DNA and RNA synthesis:
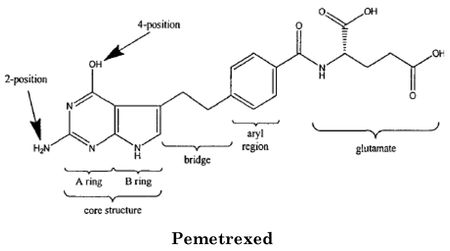
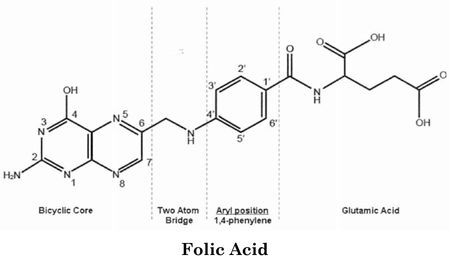
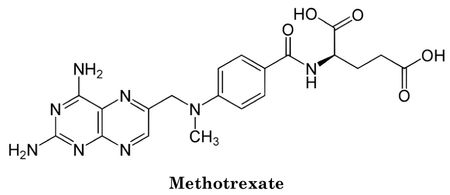
Earlier embodiments of folate inhibitors, like methotrexate, were disadvantageous because their effects on nucleic acid metabolism were felt by non-cancerous as well as cancer cells, because they inhibited cellular enzymes like dihydrofolate reductase (DHFR) that were needed for both DNA and RNA production. Pemetrexed was able to specifically inhibit DNA synthesis because it is specific for enzymes such as thymidylate synthetase (because the nucleotide thymidine is incorporated into DNA but not RNA).
Princeton University obtained (and Eli Lilly licensed) three patents related to pemetrexed: U.S. Patent No. 5,344,932 (the patent-in-suit); U.S. Patent No. 5,028,608 and U.S. Patent No. 5,248,775. The '608 patent disclosed and claimed a related but structurally distinct antifolate compound, and the '775 patent disclosed a "family of chemical intermediates that can be used to make a variety of antifolates," including pemetrexed. Important for the issues on appeal, the '775 patent claimed an intermediate in antifolate synthesis that can be used to make pemetrexed (along with a variety of other antifolate molecules). The '608 and '775 patents expired prior to the instant suit, while expiration of the '932 patent was delayed until July 14, 2016 pursuant to patent term extension under 35 U.S.C. § 156.
 Eli Lilly sued Teva, Barr Laboratories and APP Pharmaceuticals after each filed an ANDA, pursuant to 35 U.S.C. § 271(e)(2), asserting claims 1, 2, 3 and 7 of the '932 patent. At the district court, the defendants asserted as a defense that the claims of the '932 patent were invalid under the judicially established doctrine of obviousness-type double patenting over claim 3 of the '608 patent (claiming a related but structurally distinct antifolate molecule) and claim 7 of the 775 patent (that claims the intermediate). With regard to the '608 patent, Teva contended at trial that pemetrexed was related to the compound claimed by claim 3 of the '608 patent by substitution of a thienyl group with a phenyl group, something Teva asserted was "consistent with 'conventional wisdom' in the field [of the pharmaceutical arts]" and thus rendered the claims invalid. As for the '775 patent, Teva's argument was that the pemetrexed compounds claimed in the '932 patent were a "use" of the intermediate compound disclosed in the '775 patent, and were invalid for double patenting under a line of cases, the most recent of which was Sun Pharmaceuticals Industries, Ltd. v. Eli Lilly & Co. In the alternative, Teva argued that pemetrexed was merely an obvious end product for the '775 patent intermediate.
Eli Lilly sued Teva, Barr Laboratories and APP Pharmaceuticals after each filed an ANDA, pursuant to 35 U.S.C. § 271(e)(2), asserting claims 1, 2, 3 and 7 of the '932 patent. At the district court, the defendants asserted as a defense that the claims of the '932 patent were invalid under the judicially established doctrine of obviousness-type double patenting over claim 3 of the '608 patent (claiming a related but structurally distinct antifolate molecule) and claim 7 of the 775 patent (that claims the intermediate). With regard to the '608 patent, Teva contended at trial that pemetrexed was related to the compound claimed by claim 3 of the '608 patent by substitution of a thienyl group with a phenyl group, something Teva asserted was "consistent with 'conventional wisdom' in the field [of the pharmaceutical arts]" and thus rendered the claims invalid. As for the '775 patent, Teva's argument was that the pemetrexed compounds claimed in the '932 patent were a "use" of the intermediate compound disclosed in the '775 patent, and were invalid for double patenting under a line of cases, the most recent of which was Sun Pharmaceuticals Industries, Ltd. v. Eli Lilly & Co. In the alternative, Teva argued that pemetrexed was merely an obvious end product for the '775 patent intermediate.
The District Court found none of these arguments persuasive. The Court rejected Teva's "focus" on the particular portion of pemetrexed that differed from the compound disclosed in the '608 patent, saying that the weight of the evidence was that the skilled worker would have "pursued changes outside of the aryl region to improve TS inhibition and would have avoided introducing a phenyl group into the '608 Compound based on previous reports of toxicity with analogous antifolate structures." The basis for the District Court's rejection of Teva's obviousness-type double patenting argument over the disclosure of the '775 patent intermediate was that the intermediate was not claimed ("so the teachings from the '775 patent's specification were inapplicable to its obviousness-type double patenting analysis") and that there were many possible end products that could have been produced from the '775 patent intermediate and no reasoned basis for the skilled worker to have produced pemetrexed as the end product.
The Federal Circuit agreed, in an opinion by Judge Lourie joined by Judges Dyk and Wallach. The panel rejected Teva's argument on appeal, that Federal Circuit precedent (specifically, Amgen Inc. v. Hoffmann-La Roche Ltd., 580 F.3d 1340 (Fed. Cir. 2009)), mandated that a court look only at the differences between the claims at issue and the earlier claims, "so that any features held in common between the claims -- in this case, all but the aryl regions of the '608 Compound and pemetrexed -- would be excluded from consideration." The panel acknowledged that Amgen directs a court to identify the differences, but asserts that Amgen then mandates that the claims at issue be considered "as a whole" and not "the differences [] in isolation." The basis for this analytical framework is the rubric reasserted in Amgen that obviousness-type double patenting is assessed "analogous to an obviousness analysis under 35 U.S.C. §103." And as in a § 103 analysis, claims must be considered "as a whole" in an obviousness-type double patenting analysis says the panel, citing General Foods Corp. v. Studiengesellschaft Kohle mbH, 972 F.2d 1272 (Fed. Cir. 1992). "On the merits," the panel found no basis for finding clear error in the District Court's factual determinations underlying its judgment, and "absent any motivation to derive pemetrexed from the '608 Compound or reason to expect success in doing so," the panel concluded that the District Court had not erred in finding against Teva that the asserted claims of the '932 patent were not invalid for obviousness-type double patenting over the claims of the '608 patent.
Turning to Teva's obviousness-type double patenting arguments based on the teachings of the '775 patent, the panel noted that these argument's were rooted in earlier precedent, specifically citing In re Byck, 48 F.2d 665 (CCPA 1931), and Sun Pharmaceutical Industries, Ltd. v. Eli Lilly & Co., 611 F.3d 1381 (Fed. Cir. 2010). But the panel found this reliance on these cases "unsound" and proceeded to distinguish this case from that precedent. The panel began by citing the "general rule" that obviousness-type double patenting depends on a comparison with what is claimed in an earlier patent, "with the earlier patent's written description considered only to the extent necessary to construe its claims," citing In re Avery, 518 F.2d 1228, 1232 (CCPA 1975). The panel then further reaffirmed the fundamental principles that the doctrine of obviousness-type double patenting is concerned with what is claimed, not what is disclosed, because the goal of the doctrine is to prevent separate patenting of merely obvious variants of a previously patented invention.
The opinion then characterized the line of cases relied upon by Teva as "a limited exception" to this rule. The basis for this exception was set forth in Byck, wherein:
It would shock one's sense of justice if an inventor could receive a patent upon a composition of matter, setting out at length in the specification the useful purposes of such composition, manufacture and sell it to the public, and then prevent the public from making any beneficial use of such product by securing patents upon each of the uses to which it may be adapted.
Next, the Court turned to a "trio" of recent cases: Sun Pharma, Geneva Pharm., Inc. v. GlaxoSmithKline PLC, 349 F.3d 1373 (Fed. Cir. 2003), and Pfizer, Inc. v. Teva Pharm. USA, Inc., 518 F.3d 1353 (Fed. Cir. 2008). Each of these cases, according to the panel, resulted from situations where the second patent claimed a use of a composition claimed in an earlier patent that disclosed the later-claimed use. According to the opinion, these cases each "address the situation in which an earlier patent claims a compound, disclosing the utility of that compound in the specification, and a later patent claims a method of using that compound for a particular use described in the specification of the earlier patent," citing Sun Pharma, and that in each case the later application (or patent) claimed "a, or the" disclosed use of the composition claimed in the earlier patent. This analysis was enough by itself to distinguish the claims at issue here, because it was not disputed that the intermediate compound disclosed in the '775 patent (and the compound claimed in the '608 patent) were not the same compound claimed in the '932 patent, i.e., pemetrexed. Under these circumstances, "[t]he particular concerns motivating our prior decisions are [] absent here" because the claims of the '932 patent were "wholly independent" of the claims of the '775 patent. And "[p]utting aside the teachings in the '775 patent's specification, Teva's double patenting contentions evaporate." In addition, the panel noted that it had no basis for finding error in the District Court's conclusion that there was "no reason" the skilled worker would have chosen to make pemetrexed from the '775 patent intermediate, noting in passing that while not controlling, this conclusion was consistent with the provisions of the Manual of Patent Examining Procedure that states that:
[A]n intermediate product and a final product can be shown to be distinct inventions if the intermediate and final products are mutually exclusive inventions (not overlapping in scope) that are not obvious variants, and the intermediate product as claimed is useful to make other than the final product as claimed. §806.05(j).
Finally, the panel found that the District Court had erred by refusing to consider so-called "secondary" considerations of non-obviousness, on the grounds that these considerations were not to be applied in an obviousness-type double patenting assessment. But this was harmless error because the District Court had reached the correct conclusion in rejecting Teva's invalidity arguments based on obviousness-type double patenting.
The panel's opinion and discussion of the reasoning behind it appears to have cabined to a narrow category of facts the trio of decisions that applied the disclosure, as opposed to just the claims, of an earlier patent to make an obviousness-type double patenting determination on later-filed claims. Under the revised interpretation of the obviousness-type double patenting standard enunciated by the Lilly court here, the claims of an earlier patent alone are to be considered for determining obviousness-type double patenting, except insofar as the disclosure of the earlier patent is necessary to construe the scope of the earlier claims, or unless the later application claims a use for a composition claimed in an earlier patent wherein the use itself is disclosed but not claimed in the earlier patent. If this view is not further distinguished by a future panel, the Court may have brought some certainty to the application of the obviousness-type double patenting doctrine, consistent with their mandate from Congress to harmonize and clarify U.S. patent law.
Eli Lilly & Co. v. Teva Parenteral Medicines, Inc. (Fed. Cir. 2012)
Panel: Circuit Judges Lourie, Dyk, and Wallach
Opinion by Circuit Judge Lourie