The Fountain of Youth -- an enduring aspiration, particularly as the ravages of age reduce human faculties prior to leading inexorably to death. Reduction in sight is the human faculty that can have the greatest effect on quality of life in the aged -- a faculty that begins to decline in the 4th or 5th decade of life and doesn't get better (when it does) without medical intervention.
But what if there were a way to rejuvenate sight? That prospect is the tantalizing suggestion in a paper published on December 2nd entitled "Reprogramming to recover youthful epigenetic information and restore vision," Nature 588: 124-29*. The basis of the report is the recognition that many of the age-related effects on vision are an example of gene expression differences associated with epigenetic changes in chromosomal DNA. Epigenetics is a phenomenon of gene structure and expression involving small differences in nucleotide bases, typically methylation of cytosine residues at specific (CpG) sites. These changes have been studied in normal development, where gene expression changes arise as different cell types properly differentiate and act as a molecular "clock" reflecting age. The ability to turn back cellular time has been demonstrated by the development of induced pluripotent stem cells (iPSCs), wherein terminally differentiated somatic cells (typically fibroblasts) can be turned into pluripotent cells. Pluripotent cells are capable of differentiating into cells of each embryonic germinal layer (ectoderm, mesoderm, endoderm), and iPSCs can be produced by expressing four specific genes: OCT4, SOX2, KLF4 and MYC. All of these genes encode transcription factors capable of affecting (and effecting) developmentally relevant gene expression. Consequent to this "de-differentiation" occasioned by expression of these genes is a "resetting" of the epigenetic patterns associated with development. In this paper the researchers hypothesized that resetting these epigenetic patterns could also rejuvenate neuroretinal cells to reinvigorate and overcome the ocular nerve damages by glaucoma in an animal model.
Because one of these genes (MYC) is also associated with cancer development (i.e., it is an oncogene) the researchers developed an inducible expression construct that expressed only the OCT4, SOX2, and KLF4 members of the quartet (OSK). (This decision was also informed by the experience of other researchers that continuous expression of all four genes in animal models resulted in teratomas or was fatal within days of introduction.) Their system used a polycistronic (i.e., all the genes in one linear array) construct of all three genes regulated by a tetracycline response element (TRE) promoter in a adeno-associated viral vector. This construct was tested by introduction into fibroblasts from aged (20 month old) mice and gene expression related to aging (i.e., that showed differential expression with age) was evaluated. These studies showed that OSK expression for 5 days resulted in a "youthful" mRNA expression pattern in these genes (without any effect on the terminal differentiation state of the fibroblasts).
The TRE promoter enabled selection for or against expression of the OSK gene cassette; as the authors explain "[t]he TRE promoter can be activated either by reverse tetracycline-controlled transactivator (rtTA) in the presence of the tetracycline derivative doxycycline (DOX) ('Tet-On') or by tetracycline-controlled transactivator (tTA) in the absence of DOX ('Tet-Off')." Simply put, the presence of absence of DOX in the animal's drinking water determined whether the expression cassette is "on" or "off," as illustrated in this figure:
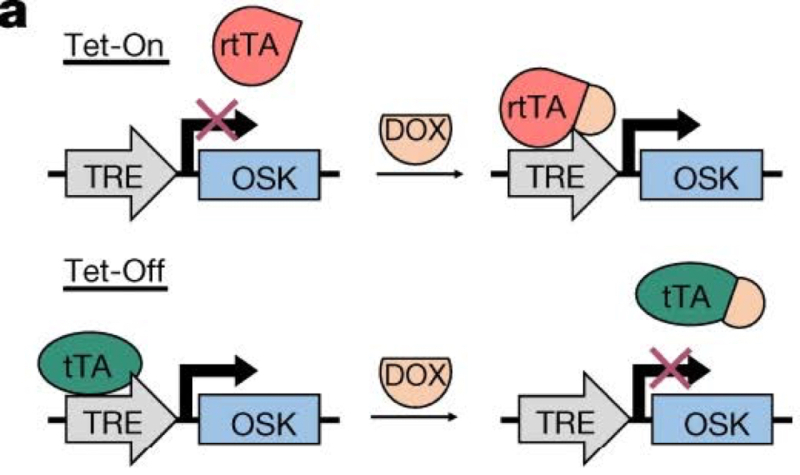
Long-term (10-18 months) expression of this cassette was achieved in both young (5 months-old) and aged mice with no tumorigenesis or other negative side effects being observed.
To test the ability of induced OSK expression to rejuvenate optical nerve cells the researchers examined retinal ganglion cells (RGC, which project axons away from the retina informing the optic nerve) in an optic nerve crush injury model (which mimics the effects of optic nerve injury due to inter alia glaucoma). The construct was delivered by injection into the vitreous humor and resulted in about 37% of the RGCs taking up and expressing the OSK genes in response to DOX administration. A separate cohort of mice were administered versions of the construct where DOX inhibited OSK expression. In these experiments, "the greatest extent of axon regeneration and RGC survival occurred when all three genes were delivered and expressed as a polycistron within the same AAV particle" according to the researchers. In contrast, inhibition of OSK expression in the "Tet-Off" mice showed no axonal growth. Moreover, delivery of the OSK genes individually in separate viral vectors or in pairs also did not show axonal growth, indicating the need for these genes to be expressed together in proper relative amounts provided by the polycistronic construct. The researchers also found OSK expression induced expression of Stat3, a gene know to encourage regeneration. These results were obtained in using 12-month-old mice as well as 1- and 3-month-old mice, which indicated, as the authors note, that "ageing does not greatly diminish the ability of OSK transcription factors to induce axon regeneration." Increased axonal growth from RGCs was found even after crush injury, an effect found with no other treatment modalities.
The researchers then determined whether these reinvigorated RGCs showed changes in DNA methylation patterns. In the absence of DOX-induced OSK expression injury in this model caused an "accelerated" aging pattern, whereas in the presence of DOX-induced OSK expression counteracted this effect according to the results reported in this paper. Interestingly, this preservation of a "youthful" pattern of DNA methylation was found to be enriched at genes "associated with light detection and synaptic transmission." Having shown this association the researchers then investigated whether axonal regeneration required youthful changes in DNA methylation. These experiments were performed by reducing expression of genes that caused DNA demethylation in RGCs (and whose expression was known to be increased in cells expressing OSK) and detecting that axonal regeneration did not occur in these mice even in the presence of DOX-induced OSK expression.
Whether these effects of OSK expression would also be seen in human neurons was investigated using differentiated human neurons in vitro. Neurons harboring an OSK-encoding construct were treated with vincristine (a drug that occasions axon injury) and DOX-induced OSK expression was shown to "counteract[] axonal loss and the advancement of DNA methylation age," showing a 15-fold greater area of proliferation in OSK-expressing cells than control vincristine-treated neural cells. These cells also showed the demethylation-dependent characteristics that were shown in RGCs in the mouse optic nerve crush injury model.
The most clinically significant result disclosed in this paper involved the effect of OSK expression in a glaucoma model in vivo. Intraocular pressure was increased to pathological levels by injecting microbeads unilaterally into the anterior chamber of mouse eye for 21 days. At 4 weeks, after these animals showed correspondingly unilateral decreases in axonal density and the number of RGCs present in the treated eye. The viral vector encoding inducible OSK expression thereafter was introduced by intravitreal injection followed by DOX-induced OSK expression for 4 weeks. Compared with control (introduction of saline or viral vectors not encoding OSK into the microbead-treated eyes) the OSK vector-treated eyes showed "restored axon density equivalent to that in the non-glaucomatous eyes, with no evidence of RGC proliferation." These mice also showed a reversal of vision loss caused by the glaucomatous injury. Together these results indicated that OSK expression could be a therapy for glaucoma in humans.
Finally, the paper reports efforts to determine whether OSK expression could improve age-related (as opposed to injury- or pathology-related) vision problems. In these experiments, 3-and 11-month-old mice were treated by intravitreal injection of DOX-inducible OSK encoding constructs and OSK expression induced for 4 weeks. Twelve-month-old mice showed age-related visual acuity and RGS electrical activity diminution which was reversed by DOX-induced OSK expression. However, these phenotypic changes were not observed to be associated with an increased number of RGCs or axon density, which prompted these researchers to hypothesize that the effect were dependent on changes in gene expression ("transcriptomic changes" as these were termed in the paper). RGCs from treated or untreated 12-month-old mice were isolated and compared with RGCs from 5-month-old mice and expression of 464 genes were found to be altered: expression of almost all (90%) of these genes were found to be restored to youthful levels in OSK-expressing RGCs. The participation of DNA methylation changes in aged RGCs in producing a youthful pattern of gene expression was further assessed and validated using artificial intelligence/machine learning approaches.
The results reported in this paper suggest therapeutic interventions that could improve vision in the aged human population even in the absence of vision-impairing pathologies such as glaucoma. Although cautious to mention that "we do not wish to imply that DNA methylation is the only epigenetic mark involved in this process" and "[i]t is likely to involve other transcription factors and epigenetic modifications," the authors are not blind to the implication that:
[W]e show that it is possible to safely reverse the age of a complex tissue and restore its biological function in vivo. Using the eye as a model system, we present evidence that the ectopic expression of OSK transcription factors safely induces in vivo epigenetic restoration of aged CNS neurons, without causing a loss of cell identity or pluripotency. Instead, OSK promotes a youthful epigenetic signature and gene-expression pattern that causes the neurons to function as though they were young again. The requirement for active demethylation in this process supports the idea that changes in DNA methylation patterns are involved in the ageing process and its functional reversal.
* By researchers from Harvard Medical School, Yale University School of Medicine, Massachusetts General Hospital, UCLA Geffen School of Medicine, and The University of New South Wales Medical School.